The excipients, that is, “biologically inert” substances added to a drug to improve their taste, color, absorption efficiency, mode and time of action in the body, are generally considered safe and harmless for those taking them, following favorable outcomes of countless tests conducted over decades and extensive use in clinical practice.
However, what is true for many people is not true for everyone. A recent research conducted in collaboration between two prestigious U.S. research centers, the Massachusetts Institute of Technology and the Brigham and Women’s Hospital – Harvard Medical School of Boston, indeed indicates that many of the theoretically “inactive” compounds contained in a tablet, capsule, effervescent powder or syrup may, in fact, be the source of intolerances, allergic reactions and hypersensitivity in susceptible patients.
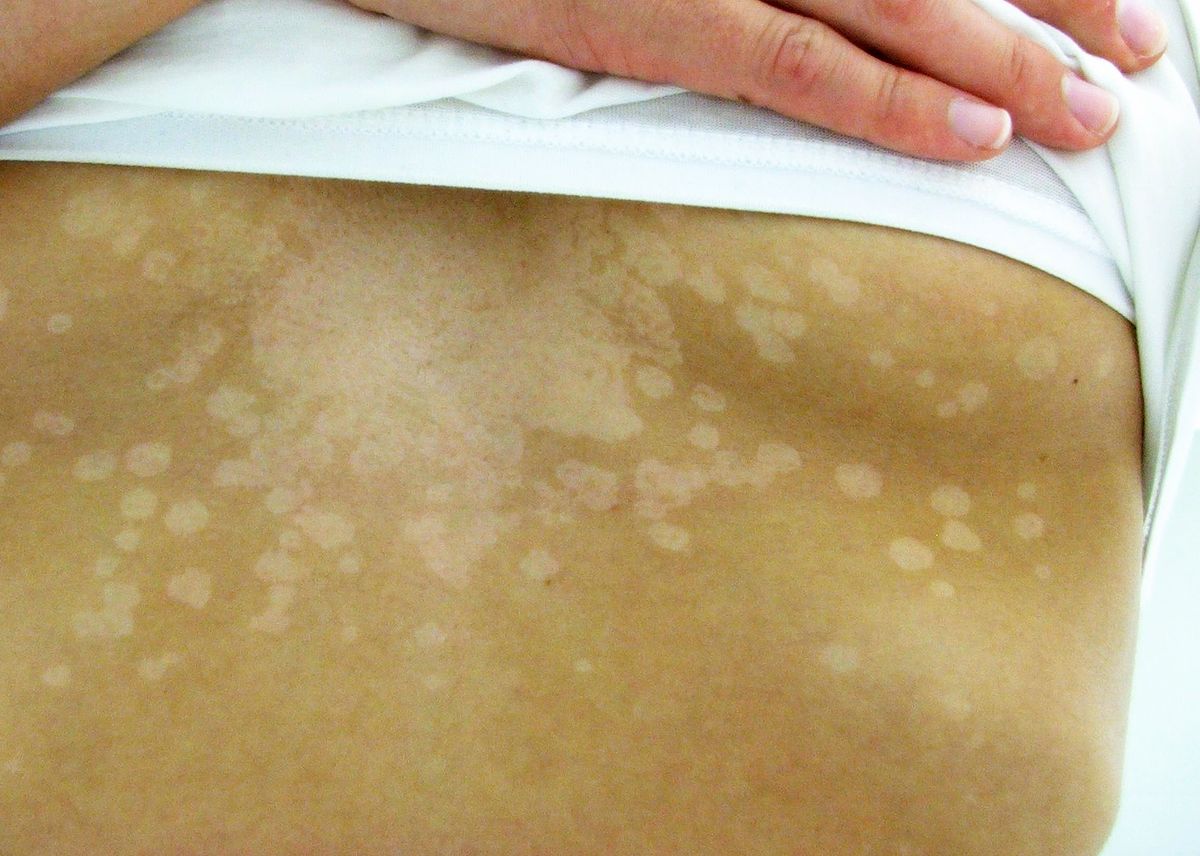
In most cases these are not serious reactions, often being limited to headaches, gastrointestinal complaints, itching or other dermatological manifestations. However, these drawbacks may lead to discontinuation or discontinuation of necessary treatment (reducing its effectiveness) or may worsen the underlying disease they are supposed to treat, especially in people who are more frail and debilitated or who have to take many different medications at once, such as the elderly.
For example, an excipient that causes gastrointestinal discomfort such as nausea or diarrhea may prevent adequate nutrition or absorption of needed nutrients in the intestines, while an “inactive” compound that induces nighttime itching may hinder rest and promote fatigue and a sense of malaise during the day.
Unfortunately, although all substances contained in a drug are listed by law in the package insert, it is not so easy for either the person taking it or the prescribing physician to have a full understanding of the associated risks, since the dosages of individual excipients are not reported and many compounds become potentially harmful only as a result of accumulation with continued use and not after one or a few intakes.
Moreover, for most of the drugs containing the same active ingredients (i.e., compounds actually endowed with therapeutic activity) there are countless different formulations containing different excipient mixes, which have further multiplied since the introduction of equivalent drugs. How to solve the situation?
According to the researchers, action needs to be taken mainly on two fronts: at the level of pharmaceutical companies, efforts should be made to develop new formulations of drugs in use, paying close attention to avoidance of excipients not necessary potentially allergenic/sensitizing (dyes, flavors, lactose, gluten, lecithins, FODMAP sugars, etc.); at the legislative level, forcing Indicate more precisely all excipients used in the package inserts.
A third and no less important aspect concerns the rational and conscious use of medications by patients, who should use medicines of any kind (over-the-counter or prescription) only in cases of actual need, respecting the directions given by the doctor or found on the package insert regarding timing and dosage, and reporting to the doctor or pharmacist any unanticipated adverse reaction, to enable more timely pharmacovigilance.
Source
Reker D et al. “Inactive” ingredients in oral medications. Science Translational Medicine 2019;11(483):eaau6753. doi10.1126/scitranslmed.aau6753 (http://stm.sciencemag.org/content/11/483/eaau6753.abstract)